|
|  |
Thermodynamics describes the molecular chaos that you find for
instance inside a steam engine. It is impossible to calculate the
positions and velocities of the each of the countless molecules in a
boiler. However, if an engineer is interested in the typical
velocities of these molecules, to determine the pressure in the boiler
for example, he can use thermodynamics. This theory provides us with
reliable calculations of such global properties by simplifying the
very complicated dynamics of the individual molecules through
statistical arguments.
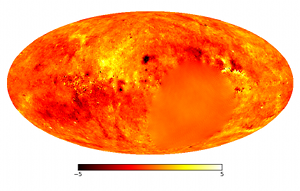
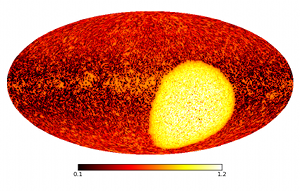
Modern astronomers collect light from outer space to produce
astronomical images in different wavelengths. At a first glance there
is not much common ground with the typical applications of
thermodynamics. When dealing with observations, however, there are
also uncertainties that can only be dealt with in a statistical
sense. The sky brightness has to be determined for theoretically an
infinite number of pixels; but the data are coarse-grained,
washed-out, noisy, most often incomplete, and always
finite. Intelligent methods are needed to convert the telescope data
into the most accurate image of the sky.
Unfortunately there are often an infinite number of possible images of
the sky to match the observational data. These possibilities are just
as confusing as the molecular chaos in a boiler but can be dealt with
using the same statistical methods. In a steam engine, the behaviour
of the water molecules is governed by two parameters: the internal
energy U of the water and its entropy S. The former is the mean energy
of the molecules due to their motion and the intramolecular attractive
forces. The latter describes the amount of molecular chaos: the larger
the entropy the more violent the motion. Thermodynamics postulates
that the water will reach an equilibrium, so that the combination of
internal energy, entropy and temperature T is minimised. This
combination is called Gibbs energy: G = U - TS, and helps to
understand water at different temperatures. At low temperatures the
water molecules want to minimise their internal energy and therefore
form drops or crystals. At high temperatures, they will form a gas,
which even though energetically costly is very chaotic (and therefore
has large entropy).
Torsten Enßlin and Cornelius Weig from the Max Planck Institute for
Astrophysics have now shown that the same terms, internal energy,
entropy and Gibbs energy, can be applied to the problem of
reconstructing digital images. The entropy describes the uncertainty
of assigning a particular brightness to the individual sky pixels. The
internal energy describes the probability of the various sky images,
which have to be taken into account within the boundaries given by the
uncertainty. The best possible sky image can then be calculated from
the interplay between internal energy and entropy. Moreover, in
contrast to traditional techniques, the new method also gives an error
map, showing the uncertainty of all pixels.
The researchers were able to show that many long established imaging
algorithms are based on this approach, which originated in the
century-old thermodynamics. However, completely new algorithms can be
developed as well. The entropy concept was already known in image
reconstruction theory, but the internal energy, which is needed to
determine the Gibbs energy, had not been introduced as such. The same
thermodynamical laws contributing to the industrial revolution could
again play an important role in today’s development of
information technologies.
Torsten Enßlin and Cornelius Weig
References:
Torsten A. Enßlin and Cornelius Weig,
"Inference with minimal Gibbs free energy in information field theory",
Phys. Rev. E 82, 051112 (2010)
 http://pre.aps.org/abstract/PRE/v82/i5/e051112 http://pre.aps.org/abstract/PRE/v82/i5/e051112
 http://arxiv.org/abs/1004.2868 http://arxiv.org/abs/1004.2868
Niels Oppermann, Henrik Junklewitz, Georg Robbers, Torsten A. Enßlin,
"Probing Magnetic Helicity in Different Astrophysical Contexts",
submitted, arXiv:1008.1246
 http://arxiv.org/abs/1008.1246 http://arxiv.org/abs/1008.1246
Taylor, A. R.; Stil, J. M.; Sunstrum, C.,
"A Rotation Measure Image of the Sky",
Volume 702, Issue 2, pp. 1230-1236 (2009)
 http://iopscience.iop.org/0004-637X/702/2/1230/ http://iopscience.iop.org/0004-637X/702/2/1230/
Links
 Research Highlight August 2009 Research Highlight August 2009
|